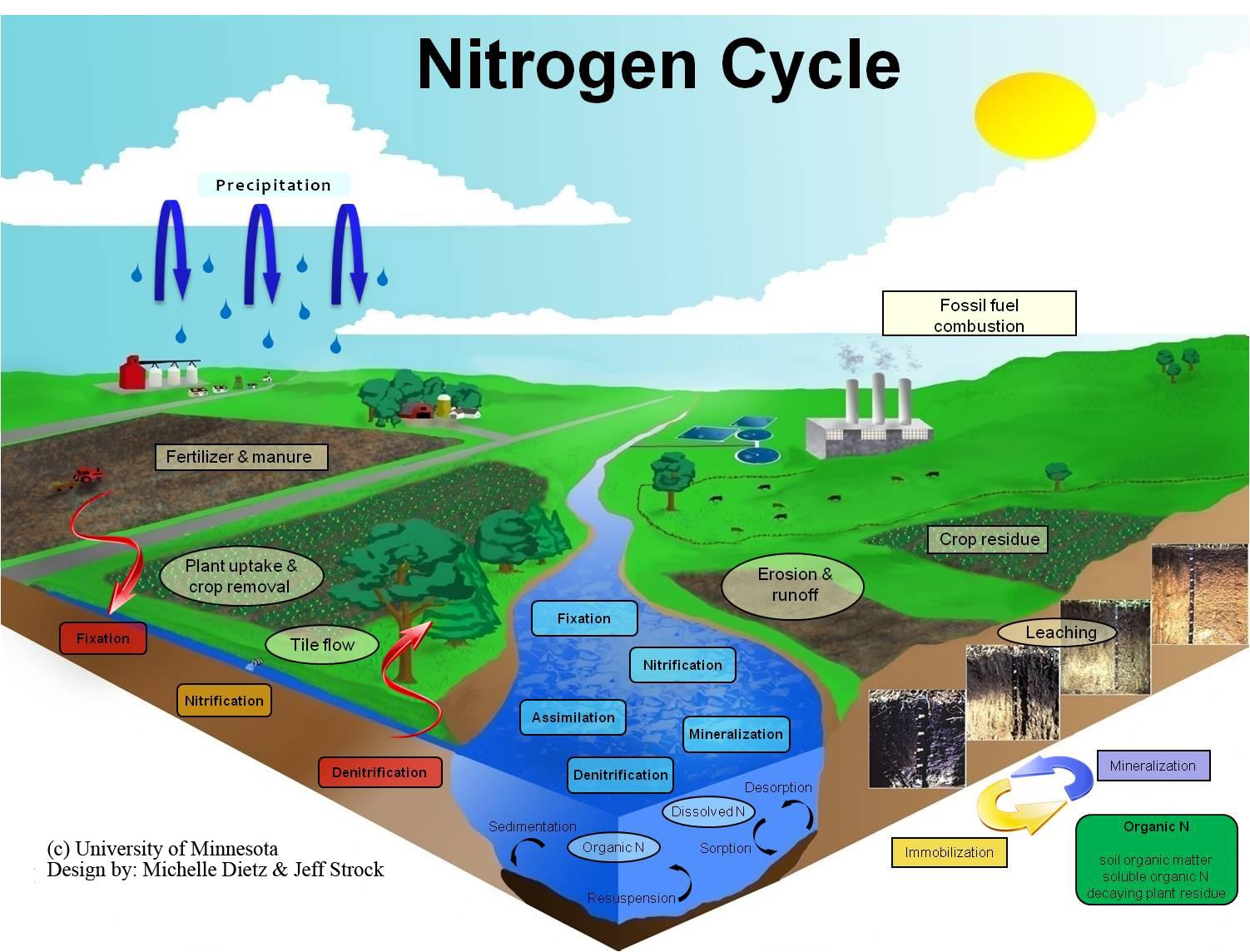
The nitrogen cycle is the biogeochemical cycle that describes the transformation and translocation of nitrogen (N) in soil, water, and living and dead organic material.
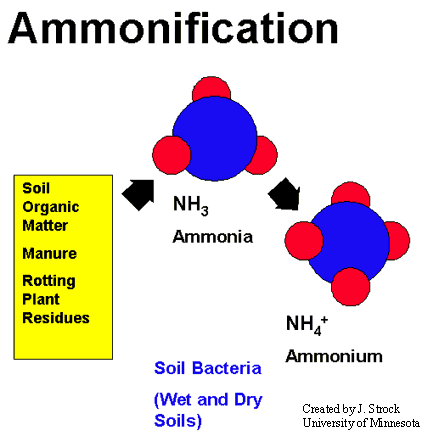
Ammonification
The biogeochemical cycling of N is highly dependent on the activities of microorganisms. Nitrogen fixation generally refers to the process in which atmospheric nitrogen (N2) is converted into ammonia (NH3). Conversion of atmospheric N to ammonia requires the enzyme nitrogenase. The conversion of organic nitrogen to NH3 is called ammonification.
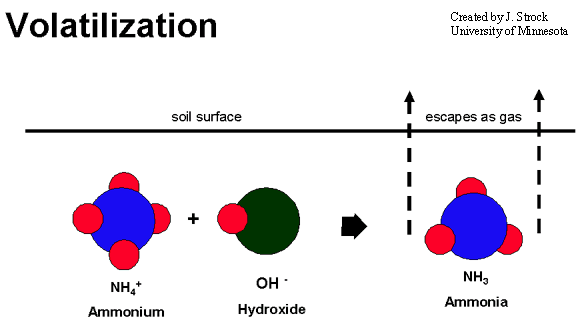
Volatilization
In the presence of water, NH3 becomes ionized and forms ammonium (NH4+). Ammonium in the soil is the starting point for a series of processes and reactions including: uptake by plants; fixation by clay minerals and organic matter; immobilization by microorganisms; transformation into ammonia gas and exported to the atmosphere by volatilization; and nitrification.
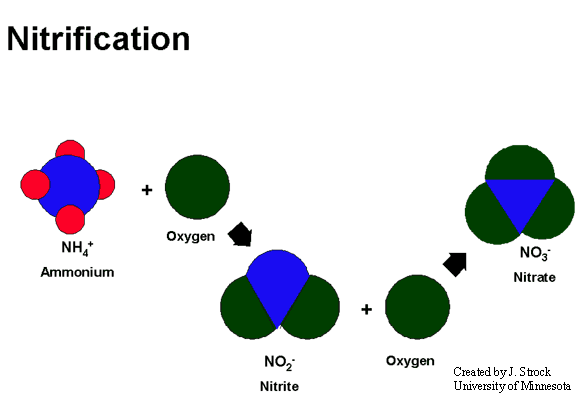
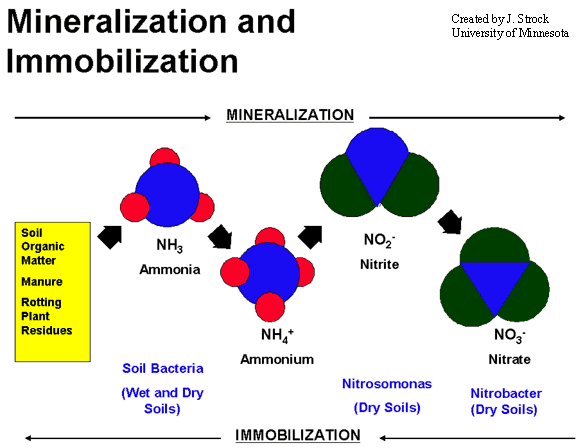
Nitrification
Nitrification is a two-step process. In the first step of this process, nitrite (NO2-) is formed when NH3 or ammonium (NH4+) is oxidized by nitrifying bacteria. Nitrifying bacteria are also responsible for the second step of the nitrification process which involves the oxidation of NO2- to nitrate (NO3-). Plants readily take up NO3- ions through their roots and assimilate them into organic compounds.
Denitrification
Denitrification is the microbially mediated process of NO3- reduction. In the absence of oxygen, NO3- ions can act as terminal electron acceptors and can result in the production of molecular nitrogen (N2) through a series of intermediate gaseous nitrogen oxide products including nitrous oxide (N2O).
Immobilization
Immobilization occurs when inorganic N, as NH4+ and NO3-, are assimilated by microorganisms. As microorganisms decompose carbonaceous organic residues they may require more N than is contained in the residue. Subsequently they assimilate inorganic N into their cellular components which becomes unavailable for plant uptake.
Mineralization
The conversion of organic N compounds to inorganic N ions is known as mineralization. In nitrogen mineralization, organic nitrogen from decaying plant and animal residues (proteins, nucleic acids, amino sugars, urea) is converted back to NH4+ and NO3-.
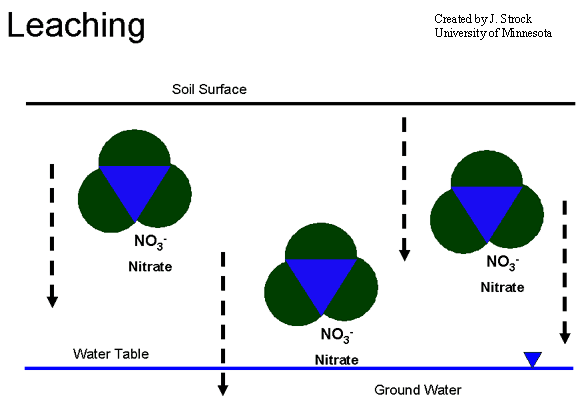
Leaching
Leaching refers to the export of N as NO3- from the soil which makes it unavailable for plant uptake. In contrast to NH4+ ions, which are attracted to negatively charged soil particles, the net negative charge of NO3- ions means that they are repelled by negatively charges soil particles. Consequently, under wet conditions, NO3- ions move downward with drainage water and are readily leached from the soil.